|
The shell following this is L shell or 2-shell. 
The shell which is closest to the nucleus is called a K shell or is represented as 1-shell. 
So again, we have a full Vaillant show between the three Astor in the three p six, which is a rocked up.An electronic shell is a part of atomic physics and chemistry, where it is an orbit which consists of electrons that are surrounding an atom's nucleus. It belongs to the noble gases with a full out to show or electron configuration has want us to to us to two p six. So, for example, neon is a P block.Įlements belongs to the noble gases are electron configuration of neon is one us to to us to to be six, so you can see that we have that full octet just here so onto the next example of are gone. Neon and argon don't have an affinity to gain or lose any electrons because they're already at the most thermodynamic lee stable state. For example, oxygen would want to gain two electrons. Often, one reactivity is driven towards attaining a full outer shell. They're often called the Frontier orbital's, so both neon and argon are in our gases, so this is because they already have a full, out of show. So these are the electrons, any very most outer shell that are involved in bonding. One more definition, before you start looking at the core material is balance electrons. This is the arrangement of our electrons in energy levels around the atomic nucleus. So electron configuration, which is also known I see electronic structure. So the first thing we'll do here is look at the definition of electron configuration, just with some contextual knowledge. Listers demonstrated how to use a period table electron configuration to identify elements. Fire as one, and it's too strong tea.Īnd that's that. We're looking at five to beginning of Krypton period. That's one that's two forty ten, which was affairs the entire block and five p two gives us a one to ten is our answer. Now begin right here with Krypton on the periodic table you go to the fifth one. We get our noble gas configuration which was all gone giving us the shorthand notations truck configuration we have for us too And greedy Tio So we'LL start back again with are very good people Then we have for us to period for that one as two and then three D too Fall in here I want you to end up with titanium is advancing next elements that were given He danced with Krypton We have fabulous tio forty two on a fall, Pete. Is that so? You may have noticed that will move into the deep lock fest is straining even though it's a period for forty, you know, in fact so that's where we'LL be again. One, two, three, four, five, six, seven, eight, nine, ten. To begin with, we're going to start, and then we move into period for as wanted us to ready. So we look at this very table games are gone. We're gonna figure out exactly what elements this is referring to a periodic table. So the first question is asking us about eh gives us a noble gas configuration first. 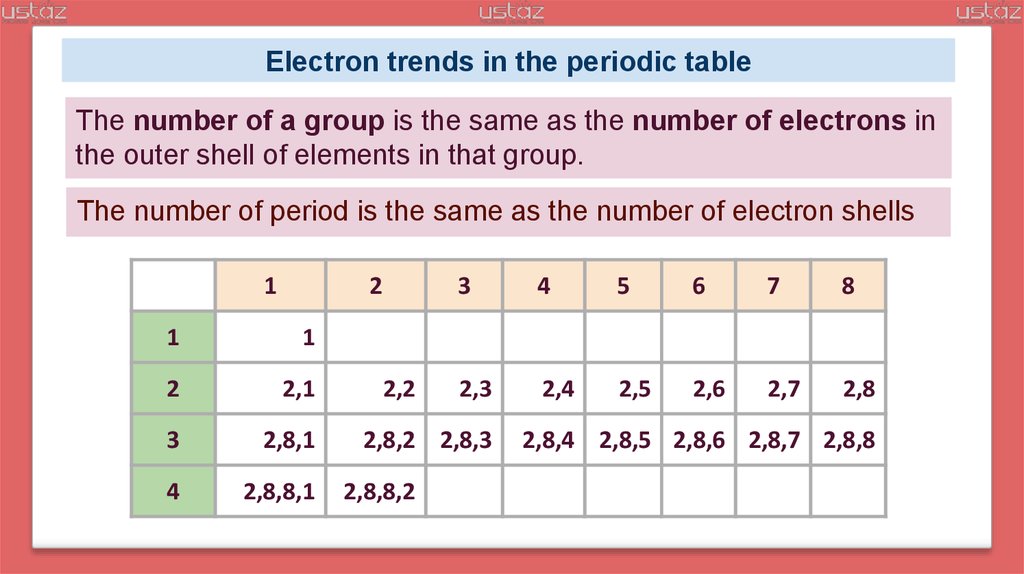
We we're not going to refer to that at this point in time that this would be your F block. A people was located here is deep.Īnd then down here we have our f block. 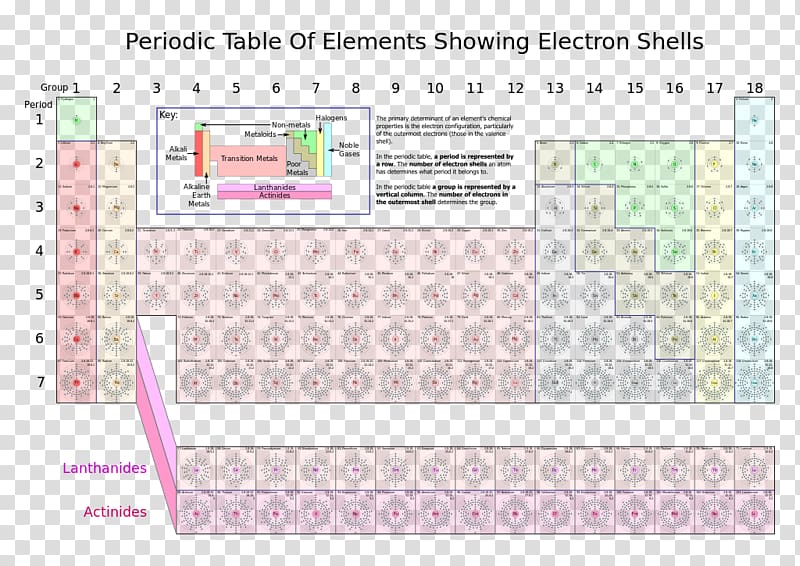
Block was located and Collins thirteen to eighteen or three a from eight. All right, Everything that's located in the first two columns is you ask. So first was briefly go with the periodic table. Chapter three problems, he says, use a periodic table to determine which element corresponds to each elect chocolate, the correction.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
